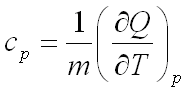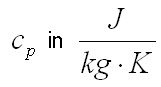Metrology
Various thermophysical variables are described on the following pages. The facilities and options available to measure thermophysical material properties within the working group can be downloaded as tables in pdf format.
Heat Capacity and Enthalpy Changes
Enthalpy changes are equal to the amount of heat a sample absorbs or releases when it is subject to a chemical or physical transition and/or temperature changes:

Specific heat capacity is the measure of the amount of heat Q required to increase the temperature of a unit quantity m of a substance by 1 K. One differentiates between specific heat under constant pressure, cp, and at constant volume cv. The following applies for specific heat under constant pressure: